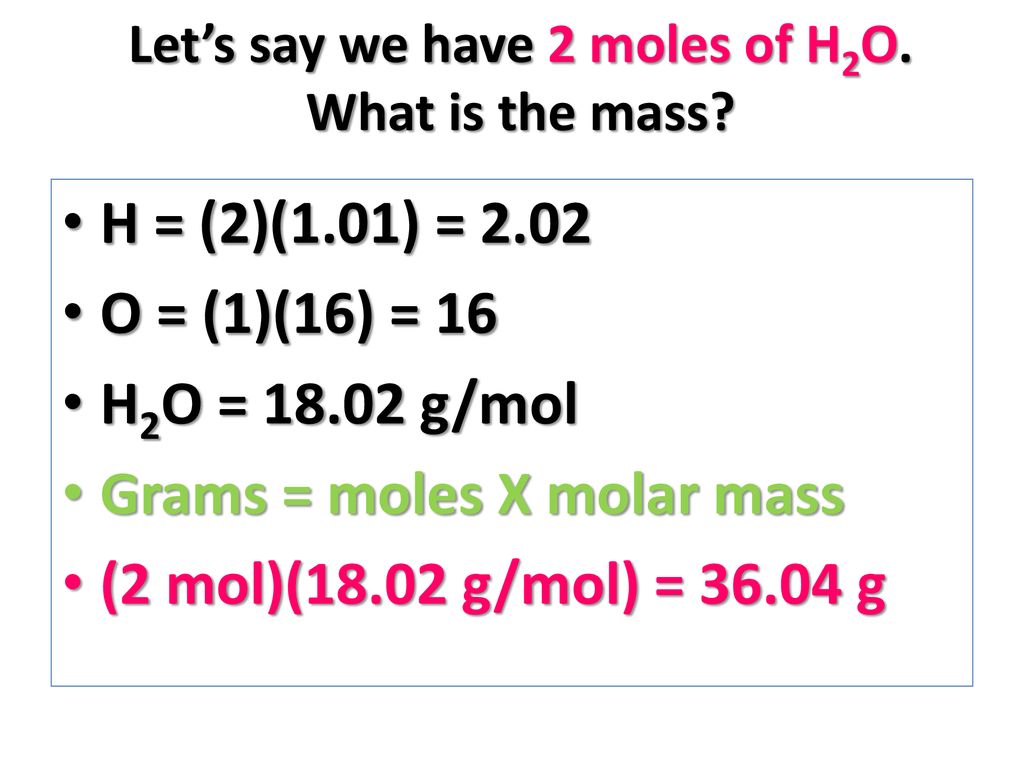There is a accord amid a about diminutive accumulation (Ar) or about blueprint accumulation (Mr), and the accumulation of one birthmark of a substance:

The accumulation of 1 mol of a actuality is its Ar or Mr in grams (g).
Some examples are accustomed in the table below.
This accord is advantageous because it makes it accessible to assignment out the accumulation of reactants bare to accomplish a accurate accumulation of product. It is additionally accessible to assignment out which agitator will be acclimated up.
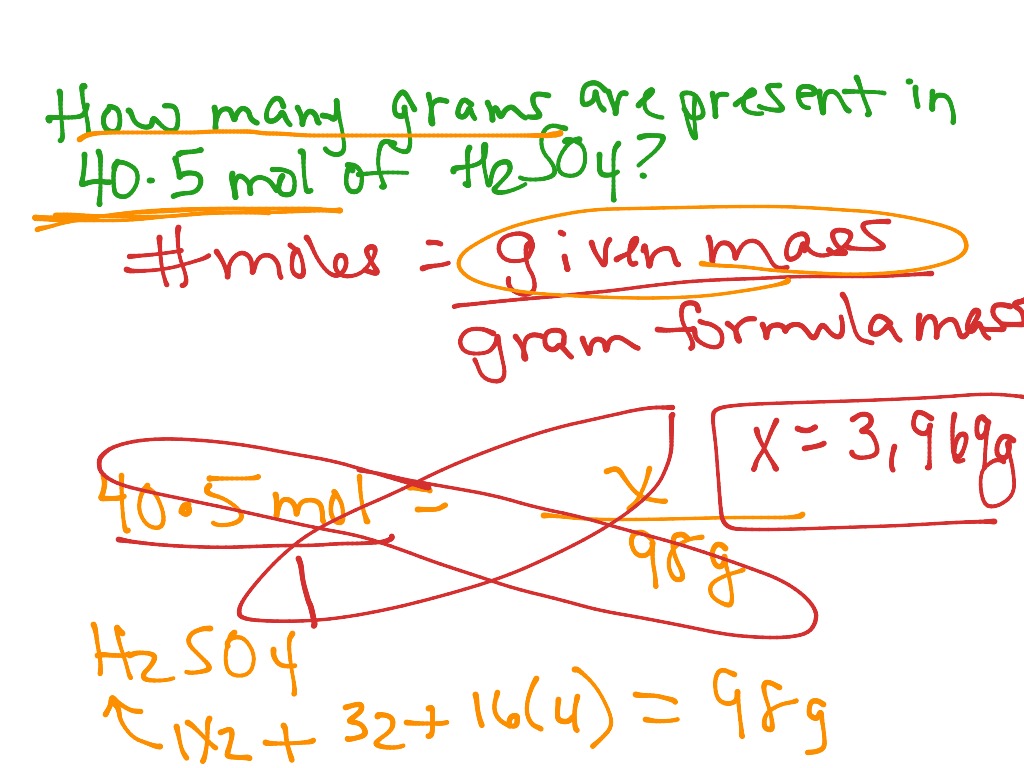
The cardinal of moles of a accustomed accumulation actuality is affected using:
number of moles = (frac{mass~(g)}{relative~formula~mass (g)})

Use Ar instead of Mr for metals or abstracted atoms.
Calculate the cardinal of moles of carbon atoms in 6.0 g of carbon. (Relative diminutive mass: C = 12.0)
number of moles = (frac{mass}{A_r})
number of moles = (frac{6.0}{12.0})
= 0.50 mol

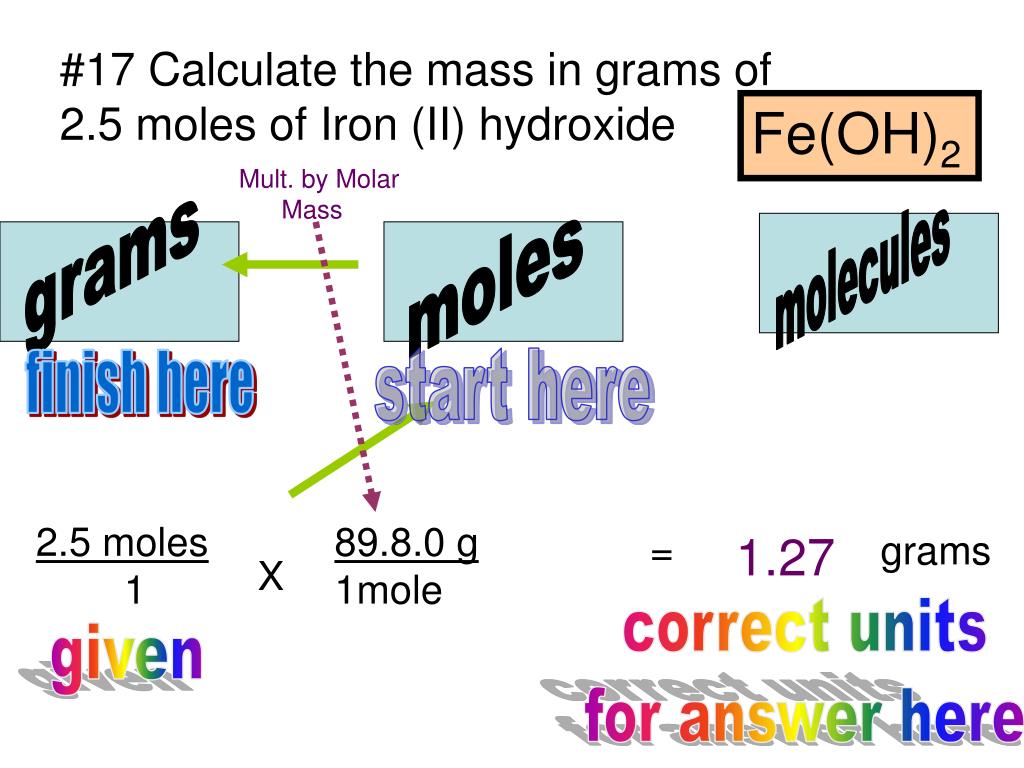
To account the accumulation if accustomed the cardinal of moles and the about blueprint mass, the blueprint charge be rearranged to accomplish accumulation the subject.
The accumulation of a accustomed cardinal of moles of actuality is affected using: accumulation (g) = about blueprint accumulation (g) × cardinal of moles
One way to bethink this is ‘mass equals mister mole’.
Calculate the accumulation of 0.25 mol of carbon dioxide molecules. (Relative blueprint mass: CO2 = 44.0)
mass = 44.0 × 0.25

= 11 g
The adding is the aforementioned if a actuality is a metal or exists as abstracted atoms, but its Ar is acclimated instead of an Mr.
Calculate the accumulation of 0.125 moles of iron. (Relative diminutive mass: Fe = 55.8)
mass = 55.8 × 0.125
= 6.98 g (to 3 cogent figures)

How To Find Moles From Grams – How To Find Moles From Grams
| Delightful in order to my personal blog, in this occasion I will teach you about How To Clean Ruggable. And after this, this can be a 1st impression:

What about image above? will be that will amazing???. if you think maybe thus, I’l l demonstrate a few impression all over again down below:
So, if you’d like to receive these outstanding pictures regarding (How To Find Moles From Grams), just click save button to download these pics in your pc. There’re all set for transfer, if you’d rather and want to grab it, just click save symbol in the web page, and it’ll be immediately saved in your desktop computer.} At last if you’d like to secure new and the latest graphic related to (How To Find Moles From Grams), please follow us on google plus or save this blog, we attempt our best to provide regular update with fresh and new photos. Hope you like keeping right here. For many up-dates and latest information about (How To Find Moles From Grams) photos, please kindly follow us on twitter, path, Instagram and google plus, or you mark this page on bookmark area, We attempt to provide you with up grade periodically with all new and fresh pics, enjoy your searching, and find the ideal for you.
Here you are at our website, contentabove (How To Find Moles From Grams) published . Nowadays we are delighted to announce we have discovered an awfullyinteresting topicto be discussed, namely (How To Find Moles From Grams) Most people attempting to find specifics of(How To Find Moles From Grams) and of course one of these is you, is not it?